  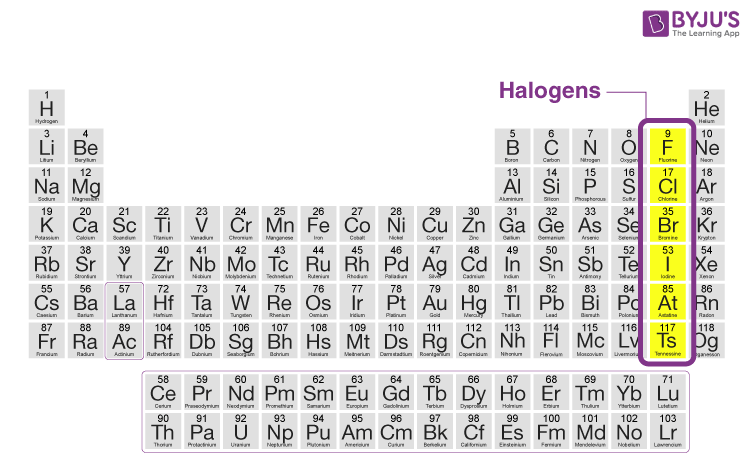
Oxyacids of halogen family elementsįluorine does not form oxy acids. The last elements of this group is tennessine(Ts),which have two synthetic radioisotopes, Ts 293 and Ts 294. These radioactive isotopes are At 215, At 217, At 218 and At 219. There are four naturally occurring radioactive isotopes of astatine which are produced via radioactive decay of uranium, neptunium, and plutonium. Several other radioactive isotopes of iodine have also been created naturally through the decay of uranium. īut ,there are trace amounts of radioactive isotope of iodine is, I 129 ,which occurs via spallation and from the radioactive decay of uranium in ores. There are no remarkable radioactive isotope of chlorine and bromine are found in nature. However, there are trace amounts of radioactive isotope of fluorine is found in nature via cluster decay of Pa231.The radioactive isotope of fluorine is F23. Fluorine has one stable natural occurring isotope, F19. Radioactive halogen family elementsĪmong the halogen family elements, fluorine, iodine and astatine have their radioactive isotope in nature. Indeed, fluorine, chlorine, bromine and iodine have closely related properties and are known collectively as the halogen family elements. Halogen family elements periodic tableĪccording to modern periodic table, group 17th consist of five elements, namely, fluorine, chlorine, bromine ,iodine and astatine which do not occur free in the nature, are collectively known as halogen family elements. Although ,the oxidizing power decrease on moving down the group. They can oxidize metal as well as non metal. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
